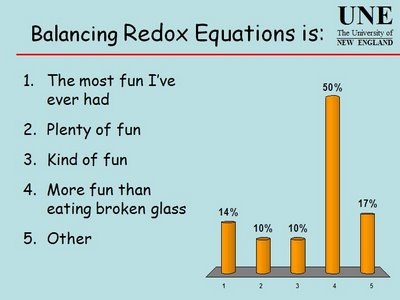
Raapa te rua tekau ma wha o Whiringa-a-nuku Thursday 24 October
Level 2 redox AS91167 (3 credits, internal)
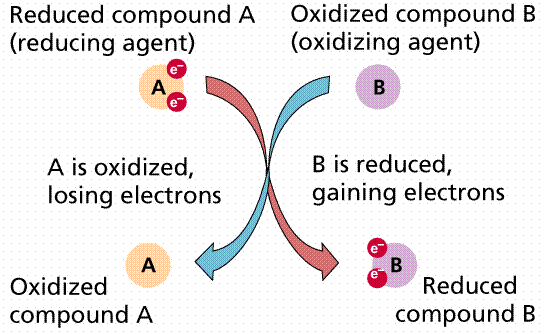
Redox reactions involve OXIDATION and REDUCTION
One reacting species is OXIDISED. This means
One reacting species is OXIDISED. This means
- LOSING ELECTRONS
- Gaining oxygen
- The species shows an increasing oxidation number (oxidation state)
- This species is the REDUCTANT (as it causes the other REACTANT species to be reduced)
The OTHER reacting species is REDUCED. This means
- GAINING ELECTRONS
- Losing oxygen/gaining hydrogen
- The species shows an decreasing oxidation number (oxidation state)
- This species is the OXIDANT (as it causes the other REACTANT species to be oxidised)
SKILL: Writing and balancing redox half equations
STEP ONE: Identify the 2 reacting species, and what they are being converted into (this is often the hardest step!)
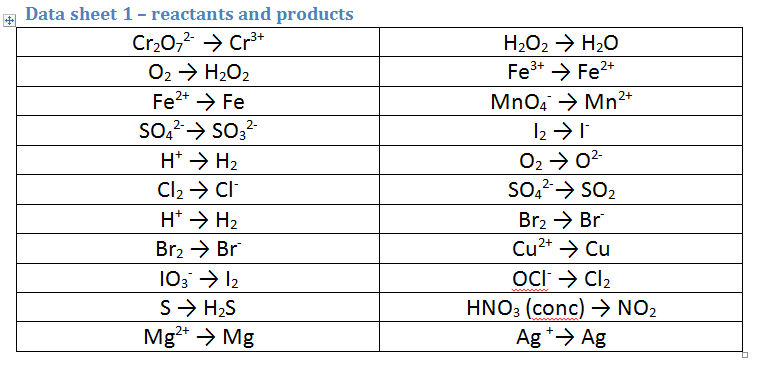
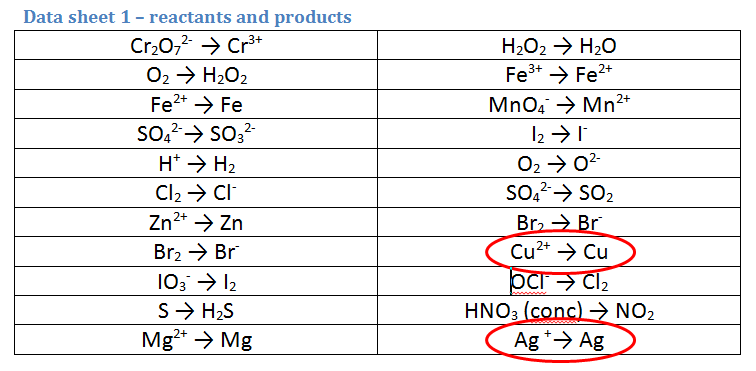
Use the data sheet (GIVEN IN THE EXAM) to identify TWO possible reactants and products for each reaction (remember one reactant gets oxidised and one gets reduced). NB these reactions CAN be reversed. Therefore you must identify TWO HALF EQUATIONS
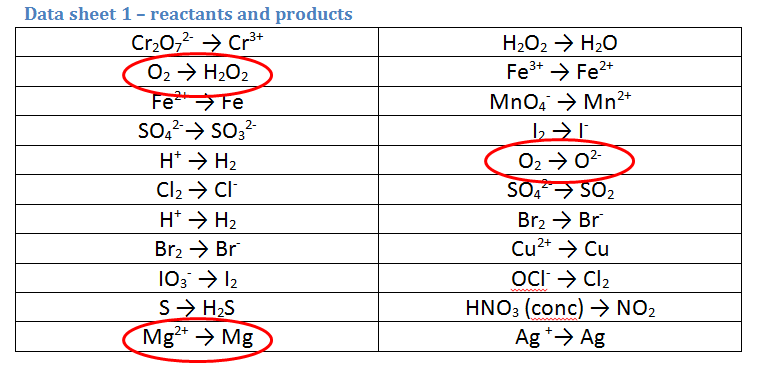
1. A piece of magnesium metal (Mg) is burnt in air (O2)
There are 3 possible equations:
There are 3 possible equations:
- There is only one equation for Mg, however it is round the wrong way as we are STARTING with Mg, so we must REVERSE this one.
- There are 2 equations for oxygen. Here, we must use our common sense to pick the most likely one. Since we have seen Mg react with oxygen (in a Bunsen burner) MANY times and NEVER have we formed H2O2 (AKA hair lightener), we must obviously choose the O2 and O2- equation for our second.
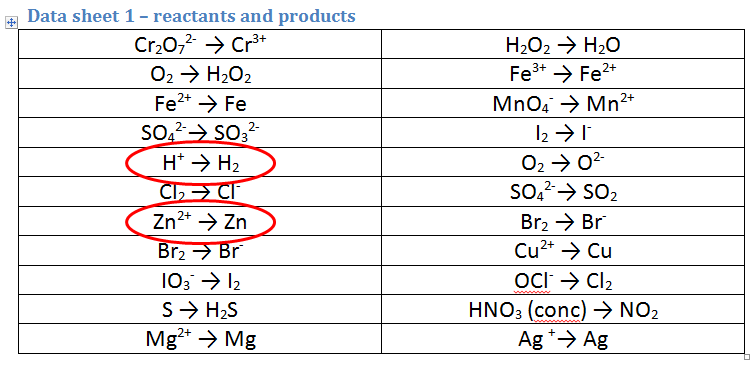
2. A piece of zinc metal (Zn) is reacted with acid (H+)
- As we are starting with acid (H+), the first equation is the correct way round. However, we are also starting with Zn metal not Zn ions, so the second equation must be REVERSED.
3. A piece of copper metal (Cu) is reacted with a solution of silver nitrate (AgNO3)
In this equation we have to analyse an ionic substance: AgNO3, which is composed of Ag+ ions and NO3- ions. We have 2 possibilities to choose from BUT ONLY ONE IS NEEDED
- Copper MUST be one of the reactants (as silver nitrate wouldn't react with itself), but the copper equation must be REVERSED so we start with Cu metal.
- The other MUST be Ag+ as there is no NO3- to choose. This equation is the right way round as we start with a SOLUTION of Ag+
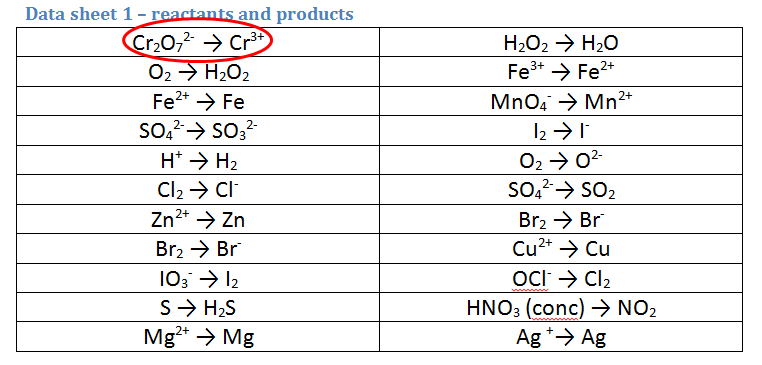
4. Solutions of acidified dichromate (Cr2O72-/H+) reacting with ethanol (C2H5OH)
- The dichromate half equation is simple to identify and is circled. It is the correct way as we do start with Cr2O72-
- This one is a bit tricky - you have to relate it back to ORGANIC CHEMISTRY!!! Oxidation of alcohols form??
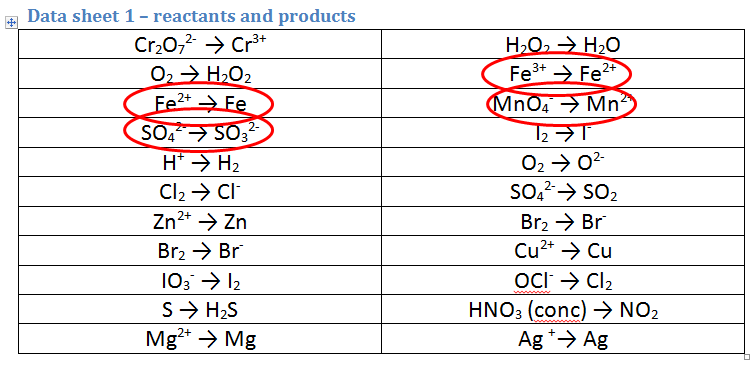
5. Solutions of acidified permanganate (MnO4-/H+) reacting with iron sulfate (FeSO4)
- The permanganate half equation is simple to identify and is circled. It is the correct way round, as we do start with MnO4-
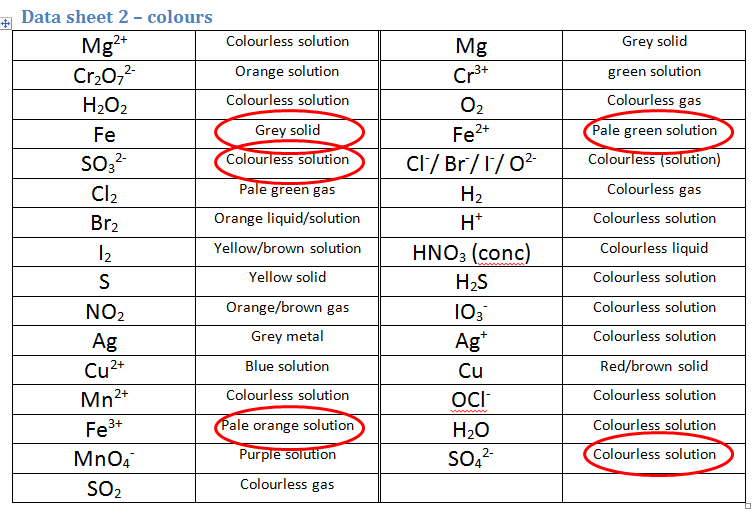
- The iron sulfate ionic solution is composed of Fe2+ ions and SO42- ions. This one is particularly tricky as we have 3 options to choose from. Usually in this case, we rely on OUR OBSERVATIONS. In this case we would observe an orange solution forming from a green solution. We can now use the second data sheet to choose the appropriate equation:
- The ONLY colours that both MATCH the observations AND are found in an equation are Fe2+ and Fe3+, therefore this must be the second correct equation.