Ramere o te iwa o Here-turi-koka Friday 8 August
Ramere o te iwa o Here-turi-koka Friday 9 August
Level 1 acids and bases AS 90944 (4 credits, external)
11SCI August 9 Aim: To get feedback!
Do now:
Get your brain and pen ready!
Get your brain and pen ready!
Aspects of acids and bases will be selected from:
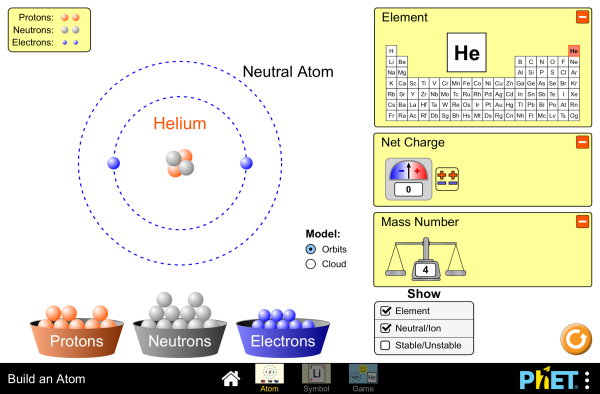
Atomic structure - electron arrangement of atoms and monatomic ions of the first 20 elements (a periodic table will be provided)
- ionic bonding
- names and formulae of ionic compounds using a given table of ions.
Properties - acids release hydrogen ions in water reactions (of acids with bases) to form salts.
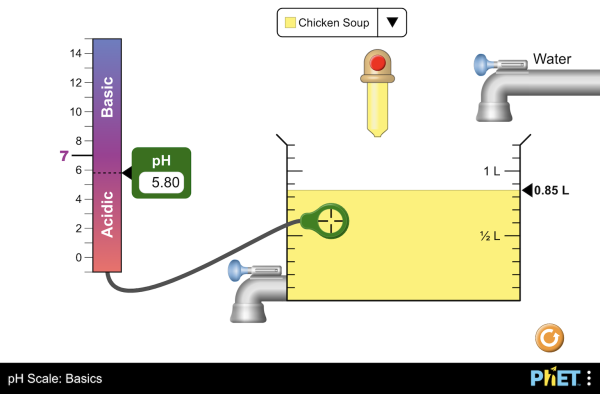
- pH and effects on indicators.
Rates of reaction and particle theory
Uses - neutralisation
- carbon dioxide formation
- salt formation.
Acids and bases are restricted to HCl, H2SO4, HNO3, metal oxides, hydroxides, carbonates and hydrogen carbonates. Other acids may be included in examination questions. The names and formulae of any such acids will be given in the question
Atomic structure - electron arrangement of atoms and monatomic ions of the first 20 elements (a periodic table will be provided)
- ionic bonding
- names and formulae of ionic compounds using a given table of ions.
Properties - acids release hydrogen ions in water reactions (of acids with bases) to form salts.
- pH and effects on indicators.
Rates of reaction and particle theory
Uses - neutralisation
- carbon dioxide formation
- salt formation.
Acids and bases are restricted to HCl, H2SO4, HNO3, metal oxides, hydroxides, carbonates and hydrogen carbonates. Other acids may be included in examination questions. The names and formulae of any such acids will be given in the question
| pdf_notes_ncea_science_1.5__gz_.pdf | |
| File Size: | 4022 kb |
| File Type: | |
|
Here's a clip showing what elements in the SAME GROUP (column) have in common - all have the same number of valence electrons, form ions with the same charge and react in a similar way!
|
Check out this clip on rates of reaction!
|