Rahina te rua tekau matoru o Poutu-te-rangi Monday 23 March
level 3 Bonding and Thermochemistry
as91390 (5 credits, external)
|
|
|
SCIPAD PAGES - Bonding
|
|
13 CHEM Bonding and thermochem test feedback 2015:
1. learn e configuration (and don't forget 4s = first in and first out)
2. annotate periodic trends with #protons and electron shells (and then use these in your answer)
3. less flowery language - even bullet points would be preferable
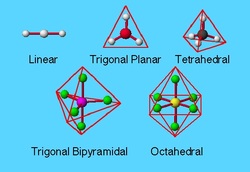
4. learn your shapes and nail your molecular polarity question - no excuses!
5. learn the definitions for the types of enthalpy change
6. read the question carefully - annotate with molar mass, types of WIF etc
7. consider the polarity of the molecule does not equal explain why the molecule is polar
8. "more bonds" is not a reason for a difference in enthalpy change
9. the bullet points help you make sure you have included all the key points BUT THE BULLET POINTS ARE NOT THE QUESTION! You must answer the question
10. Get the simple things right - eg exothermic = DH- = heat released
11. ALL chemical reactions break AND form bonds!
12. Molecules are molecular and form WIF BETWEEN MOLECULES. Molecules do NOT form ionic bonds.
13. You have learned 3 types of WIF. There is 100% chance of you having to use these in your exam.
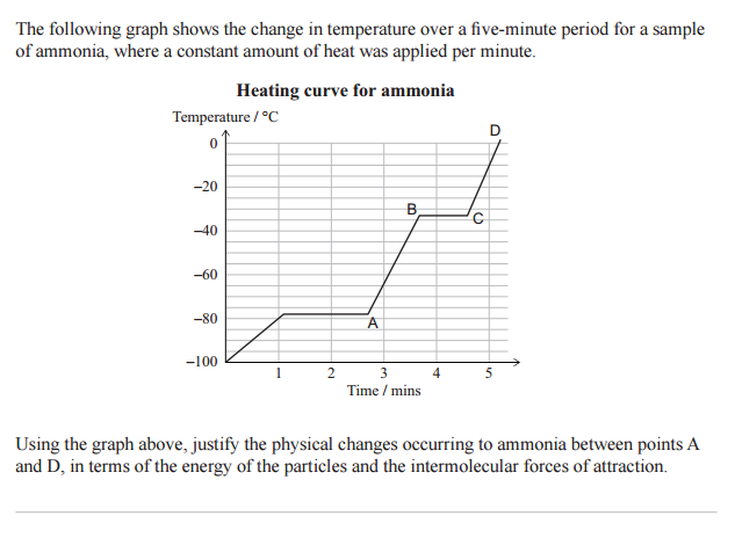
14. You must be able distinguish between a chemical reaction and a physical change (eg boiling, melting).
15. In physical changes like boiling and melting YOU DO NOT break the covalent bonds between atoms!!!
16. K.I.S
1. learn e configuration (and don't forget 4s = first in and first out)
2. annotate periodic trends with #protons and electron shells (and then use these in your answer)
3. less flowery language - even bullet points would be preferable
4. learn your shapes and nail your molecular polarity question - no excuses!
5. learn the definitions for the types of enthalpy change
6. read the question carefully - annotate with molar mass, types of WIF etc
7. consider the polarity of the molecule does not equal explain why the molecule is polar
8. "more bonds" is not a reason for a difference in enthalpy change
9. the bullet points help you make sure you have included all the key points BUT THE BULLET POINTS ARE NOT THE QUESTION! You must answer the question
10. Get the simple things right - eg exothermic = DH- = heat released
11. ALL chemical reactions break AND form bonds!
12. Molecules are molecular and form WIF BETWEEN MOLECULES. Molecules do NOT form ionic bonds.
13. You have learned 3 types of WIF. There is 100% chance of you having to use these in your exam.
14. You must be able distinguish between a chemical reaction and a physical change (eg boiling, melting).
15. In physical changes like boiling and melting YOU DO NOT break the covalent bonds between atoms!!!
16. K.I.S
|
Click HERE for some good (but annoying) explanations of molecular shape
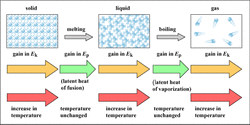 Who knew that the boiling of water could be so complicated? Who knew that the boiling of water could be so complicated?
|
|
|
Some basic notes on polarity, Lewis diagrams, WIF and periodic trends (thanks to No Brain is Too Small)
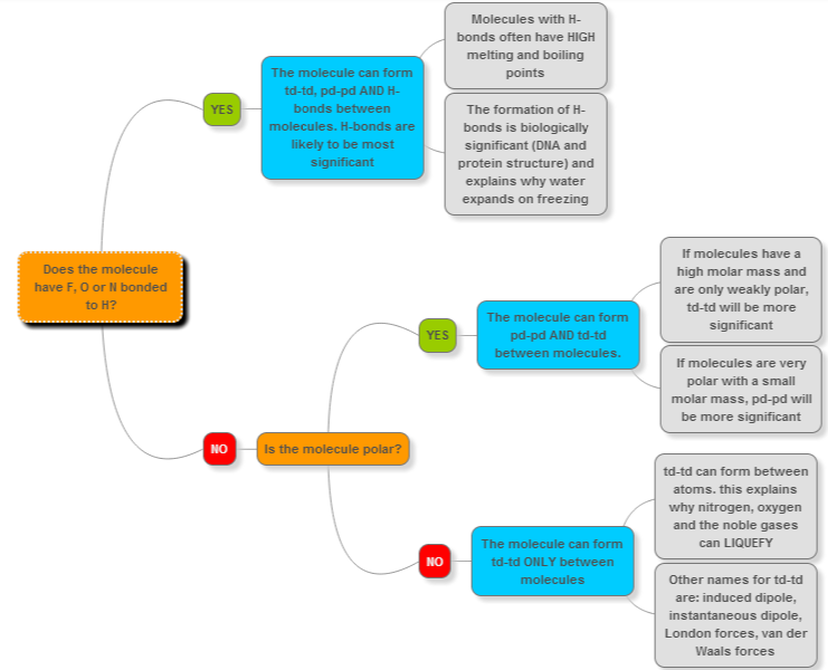
Check out this decision tree I made for deciding what types of WIF are present between molecules. You can make your own using Google Apps if you have a gmail address.
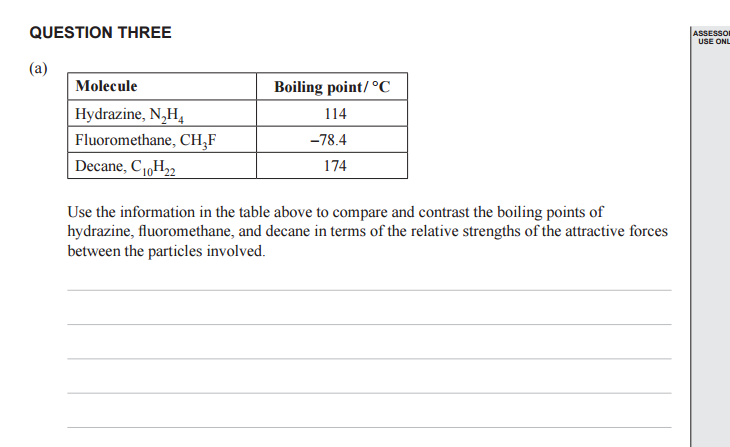
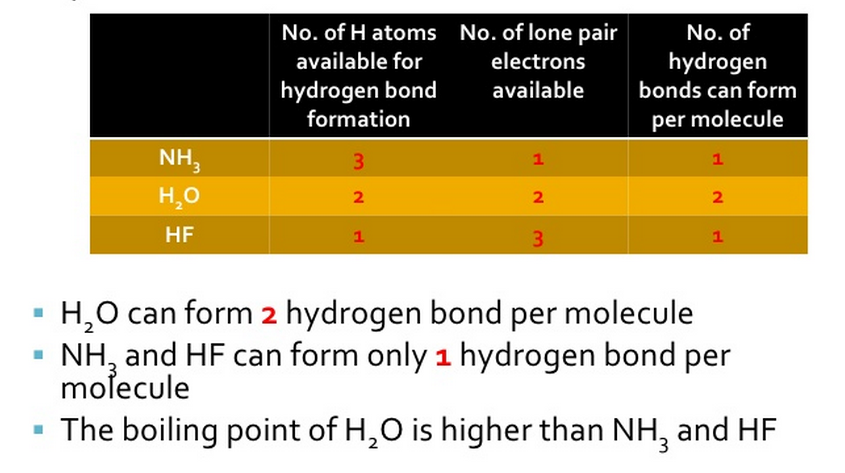
The table below looks in detail to hydrogen bonding and explains the differences in boiling points of 3 molecules that can all form H-bonds
Enthalpy notes and worksheets
|
|
|
Entropy notes
|
|
|