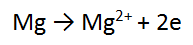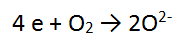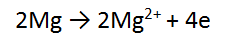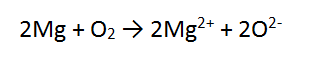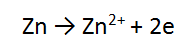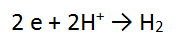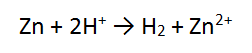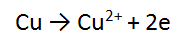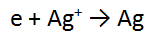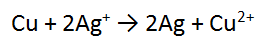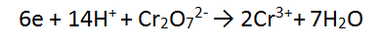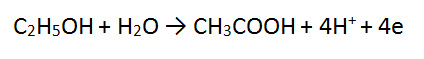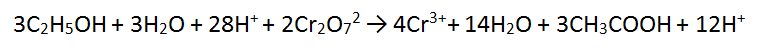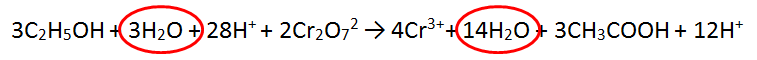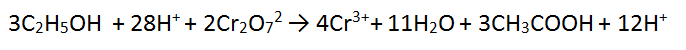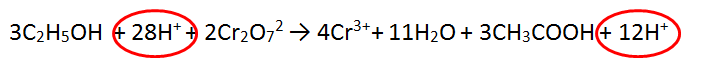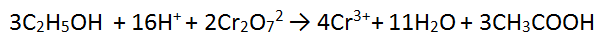STEP 3: ADDING THE HALF EQUATIONS TOGETHER
1. A piece of magnesium metal (Mg) is burnt in air (O2)
You now have 2 balanced half equations:
You now have 2 balanced half equations:
To add these together, you must MAKE SURE THE NUMBER OF ELECTRONS GAINED EQUALS THE NUMBER OF ELECTRONS LOST.
In this case, the numbers are NOT equal, therefore we must multiply one or both equations so that they are equal.
Multiply ALL of equation one by 2
In this case, the numbers are NOT equal, therefore we must multiply one or both equations so that they are equal.
Multiply ALL of equation one by 2
Add the 2 half equations together. No, it does not matter which order the reactants are written or the products. The electrons CANCEL OUT and are not written in the final answer:
2. A piece of zinc metal (Zn) is reacted with acid (H+)
In this case, the number of electrons is the same in both equations, so we can just add these as they are, cancelling the electrons.
3. A piece of copper metal (Cu) is reacted with a solution of silver nitrate (AgNO3)
In this case, we must multiply the second equation by 2, and then add them cancelling the electrons.
4. Solutions of acidified dichromate (Cr2O72-/H+) reacting with ethanol (C2H5OH)
NB this one is tricky, skip to number 5 if you like!
NB this one is tricky, skip to number 5 if you like!
In this tricky case, we must multiply BOTH equations - we can use the lowest common multiple which is 12. Therefore we multiply equation 1 by 2 and equation 2 by 3. This makes both equations have equal electrons. In this case there is MORE CANCELLING to be done.
1. Cancel 3 waters from each side
2. Cancel 12 H+ from each side
FINAL ANSWER! This is an excellence skill