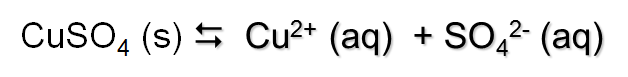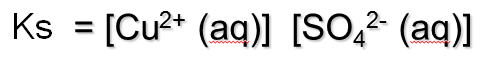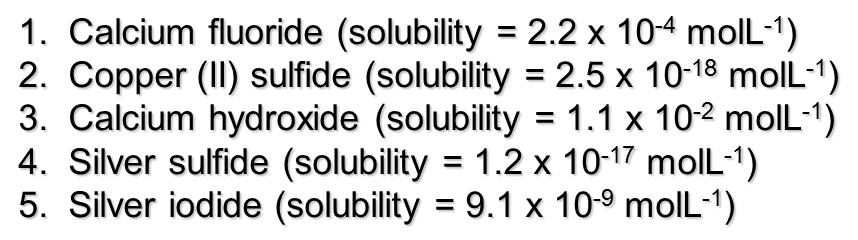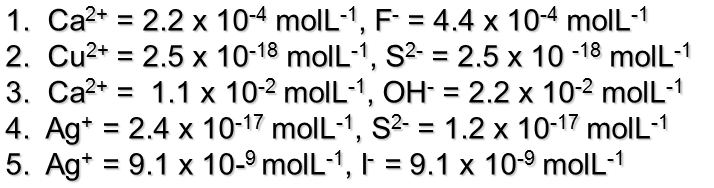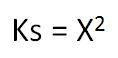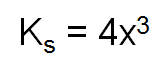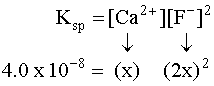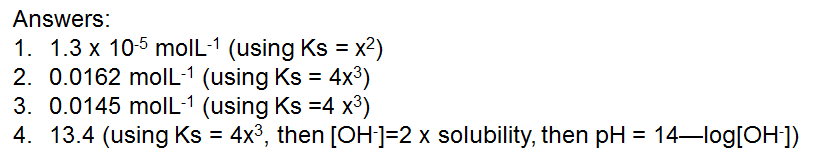Ratu te tekau ma rima o Whiringa-a-nuku Tuesday 15 October
PART 2: SOLUBILITY
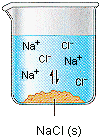
- Solubility is the amount of a substance (solute) that can dissolve in a solvent (water) at a certain temperature to make a solution
- Solubility can be measured in g/L or mol/L
- A saturated solution is one in which no further solid can dissolve (at that temperature)
Consider the situation where solid copper sulfate is dissolved in water
As you add solid CuSO4 to water, it dissolves until it reaches the saturation point. This is the point at which no more solid will dissolve. You know when you’ve reached saturation when there is undissolved solid in the beaker.
At this point an equilibrium system is established where the rates of the forward and reverse reactions are equal.
A K expression can be written for this equilibrium, remembering that solids do not have a concentration, so are not included in the expression. Ks = SOLUBILITY PRODUCT.
The Ks is the SOLUBILITY PRODUCT – the product of the concentration of ions in a saturated solution of a sparingly soluble compound. Did I mention that the Ks is the solubility product? Ks = solubility product.
TRY THIS: Use the solubility to calculate the concentration of ions in the following solutions:
Answers:
CALCULATING Ks FROM SOLUBILITY
Very simply, if the compound is a 1:1 compound (where the ions are in a 1:1 mole ratio eg NaCl), then
Where x = solubility in moles per litre
If the compound is a 1:2 (or 2:1) mole ratio eg CaCl2 or K2O, then
If the compound is a 1:2 (or 2:1) mole ratio eg CaCl2 or K2O, then
Again, x= moles per litre. (NB: you should understand this better, check with your teacher for the understanding)
Try these :
Try these :
Answers to the above:
CALCULATING solubility FROM Ks (solubility product)
You must obviously mathematically reverse the previous formulae: (square root or divided by 4 and then cube root)
Try these: (answers in molL-1 or pH)
Try these: (answers in molL-1 or pH)
Answers:
Try these collated NCEA questions on Solubility basics