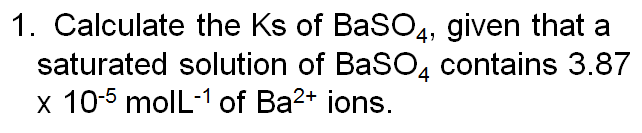Te Ramere o te tekau ma wha o Whiringa-a-nuku Friday 14 October
13 Chem do now:
a) write the formulae for the following: propylethanoate, 2-chloropentanoic acid, trichloromethane, an amino acid with a side chain of an H atom and b) choose the molecule with a chiral C and draw 3D representations of the enatiomers it forms
a) write the formulae for the following: propylethanoate, 2-chloropentanoic acid, trichloromethane, an amino acid with a side chain of an H atom and b) choose the molecule with a chiral C and draw 3D representations of the enatiomers it forms
Today's Aim: To practise questions on the common ion effect and practise using it and to remember complex ions and consider their effect on solubility
IONIC PRODUCT
The ionic product is the product of the ions (according to the Ks expression). It looks like the Ks! However there is one important difference:
Try these questions to see the difference:
- In a Ks expression, the [ions] comes from the dissociation of the salt (in a SATURATED solution), therefore the ratio of ions is constant: 1:1, 2:1 or 1:2
- In an ionic product, the ratio of ions is not constant - i.e. each ion can come from a different source, and therefore the concentration of each ion is independent of the other
Try these questions to see the difference:
Answers:
Using the Ionic Product to predict precipitation
In the case above, the water supply would form a precipitate as the IP>Ks.
Now try these collated NCEA questions on predicting precipitation.
HINT: start with the substance you have been given the Ks for and write a Ks expression for it
- If the ionic product is GREATER than the Ks, then the solution is PAST SATURATION and a PRECIPITATE forms.
- If the ionic product is LESS than the Ks, then the solution is NOT YET SATURATED and NO PRECIPITATE forms.
In the case above, the water supply would form a precipitate as the IP>Ks.
Now try these collated NCEA questions on predicting precipitation.
HINT: start with the substance you have been given the Ks for and write a Ks expression for it