Te Ramere o te tekau ma wha o Whiringa-a-nuku Friday 14 October
12 CHEM Do Now: Calculate the concentration of H3O+ and OH- in the following solutions: a) 0.00435 molL-1 HCl and 0.0667 molL-1 NaOH. State any assumptions you have made.
12Chem aim: to put it all together and do some FULL practices for the assessment
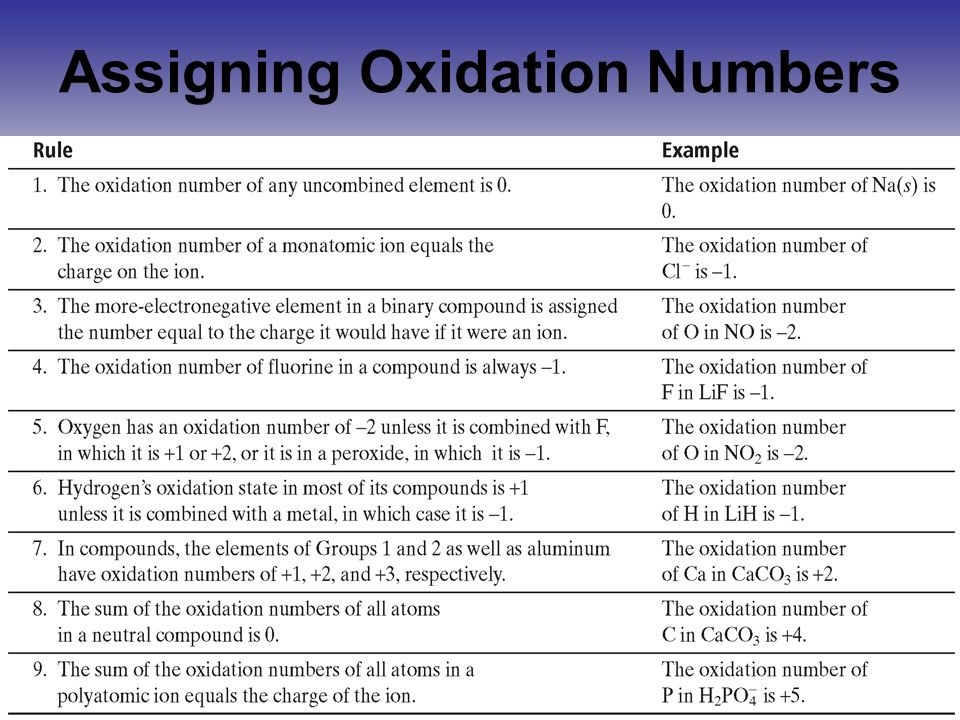
ASSIGNING OXIDATION NUMBERS
- Oxidation numbers are assigned to ELEMENTS (not compounds)
- YOU DO NOT have to know how to assign and use oxidation numbers - you can choose to deal in electron transfers; HOWEVER:
- If you are considering carrying on to Level 3 it is useful to learn this and pretty much imperative if you are considering scholarship or university
- If an element increases oxidation number it shows OXIDATION
- If an element decreases oxidation number it shows REDUCTION
Have a go at this worksheet: