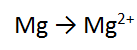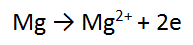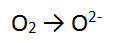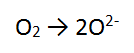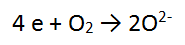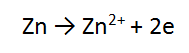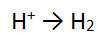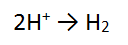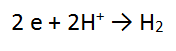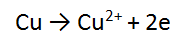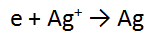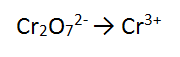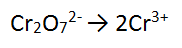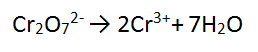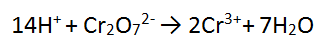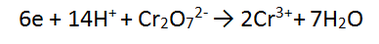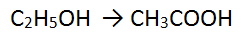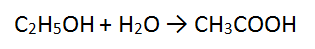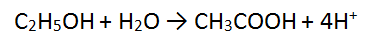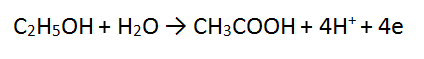STEP 2: BALANCING THE HALF EQUATIONS
1. A piece of magnesium metal (Mg) is burnt in air (O2)
REACTANT AND PRODUCT ONE:
REACTANT AND PRODUCT ONE:
FOLLOW THE STEPS!!!!
Step 1: There is one Mg on each side = balanced
Step 2: There are no O atoms to balance = done
Step 3: There are no H atoms to balance = done
Step 4: The most positive side is the RIGHT, it is more positive by 2, therefore add 2 e to the RHS = DONE!!
Step 1: There is one Mg on each side = balanced
Step 2: There are no O atoms to balance = done
Step 3: There are no H atoms to balance = done
Step 4: The most positive side is the RIGHT, it is more positive by 2, therefore add 2 e to the RHS = DONE!!
REACTANT AND PRODUCT TWO:
FOLLOW THE STEPS!!!!
Step 1: There are no non H or O atoms = done
Step 2: There are 2 O atoms on the left and only 1 on the right so balance the O's = done
Step 1: There are no non H or O atoms = done
Step 2: There are 2 O atoms on the left and only 1 on the right so balance the O's = done
Step 3: There are no H atoms to balance = done
Step 4: The most positive side is the LEFT, it is more positive by 4, therefore add 4 e to the LHS = DONE!!
Step 5: check that one equation has electrons (e) on the LHS and the other on the RHS. IF NOT: Mistakes have been made!
Step 4: The most positive side is the LEFT, it is more positive by 4, therefore add 4 e to the LHS = DONE!!
Step 5: check that one equation has electrons (e) on the LHS and the other on the RHS. IF NOT: Mistakes have been made!
2. A piece of zinc metal (Zn) is reacted with acid (H+)
REACTANT AND PRODUCT ONE:
Follow the same steps for Zn as you did for Mg
Follow the same steps for Zn as you did for Mg
REACTANT AND PRODUCT TWO:
FOLLOW THE STEPS!!!!
Step 1: There are NO non H or O atoms = done
Step 2: There are no O atoms to balance = done
Step 3: There are 2 H atoms on the RHS and only 1 on the LEFT so must balance
Step 1: There are NO non H or O atoms = done
Step 2: There are no O atoms to balance = done
Step 3: There are 2 H atoms on the RHS and only 1 on the LEFT so must balance
Step 4: The most positive side is the LEFT, it is more positive by 2, therefore add 2 e to the LHS = DONE!!
Step 5: check that one equation has electrons (e) on the LHS and the other on the RHS. IF NOT: Mistakes have been made!
Step 5: check that one equation has electrons (e) on the LHS and the other on the RHS. IF NOT: Mistakes have been made!
3. A piece of copper metal (Cu) is reacted with a solution of silver nitrate (AgNO3)
REACTANT AND PRODUCT ONE:
Follow the same steps for Cu as you did for Mg and Cu
Follow the same steps for Cu as you did for Mg and Cu
REACTANT AND PRODUCT TWO:
Remember: you have NOT started with Ag metal, but Ag+ ions in solution
Remember: you have NOT started with Ag metal, but Ag+ ions in solution
4. Solutions of acidified dichromate (Cr2O72-/H+) reacting with ethanol (C2H5OH)
NB this one is tricky, skip to number 5 if you like!
NB this one is tricky, skip to number 5 if you like!
REACTANT AND PRODUCT ONE:
FOLLOW THE STEPS!!!!
Step 1: There is one Cr on the RHS and 2 on the LHS - need to balance
Step 1: There is one Cr on the RHS and 2 on the LHS - need to balance
Step 2: There are 7 O atoms on the LHS - add 7 water to the RHS to balance the O atoms
Step 3: There are 14 H atoms on the RHS so add 14H+ to the LHS to balance
Step 4: There are a total of 12 + on the LHS and 6 + on the RHS. The LHS is the most positive by 6, so add 6 e to the LHS to balance
REACTANT AND PRODUCT TWO:
FOLLOW THE STEPS!!!!
Step 1: There are 2 C atoms on each side - balanced
Step 2: There are 2 O atoms on the RHS and 1 on the LHS - balance using water
Step 1: There are 2 C atoms on each side - balanced
Step 2: There are 2 O atoms on the RHS and 1 on the LHS - balance using water
Step 3: There are 8 H atoms on the LHS and 4 H atoms on the RHS so add 4H+ to the RHS to balance
Step 4: The most positive side is the RHS, it is more positive by 4, therefore add 4 e to the RHS = DONE!!
Step 5: check that one equation has electrons (e) on the LHS and the other on the RHS. IF NOT: Mistakes have been made!
Step 5: check that one equation has electrons (e) on the LHS and the other on the RHS. IF NOT: Mistakes have been made!